This article is part of a series in The Hospital Leader written by members of the Division of Hospital Medicine at Dell Medical School at The University of Texas in Austin, exploring lessons learned from the coronavirus pandemic and outlining an approach for creating COVID-19 Centers of Excellence.
Evidence on emerging treatments for COVID-19 has been incomplete, often disappointing, and rapidly changing. The concept of a practice-changing press release is as novel as the coronavirus. The pandemic has created an interdependent set of inpatient challenges: keeping up with evolving science and operationalizing clinical workflows, technology, and therapeutics to adapt what we are learning. At Dell Medical School, we have created a Therapeutics and Informatics Committee to put evidence into practice in real-time, and below is a brief framework of what we have learned to date:
The COVID-19 disease course can be broken down into 3 stages, and workup and interventions should be targeted to those stages1–3.
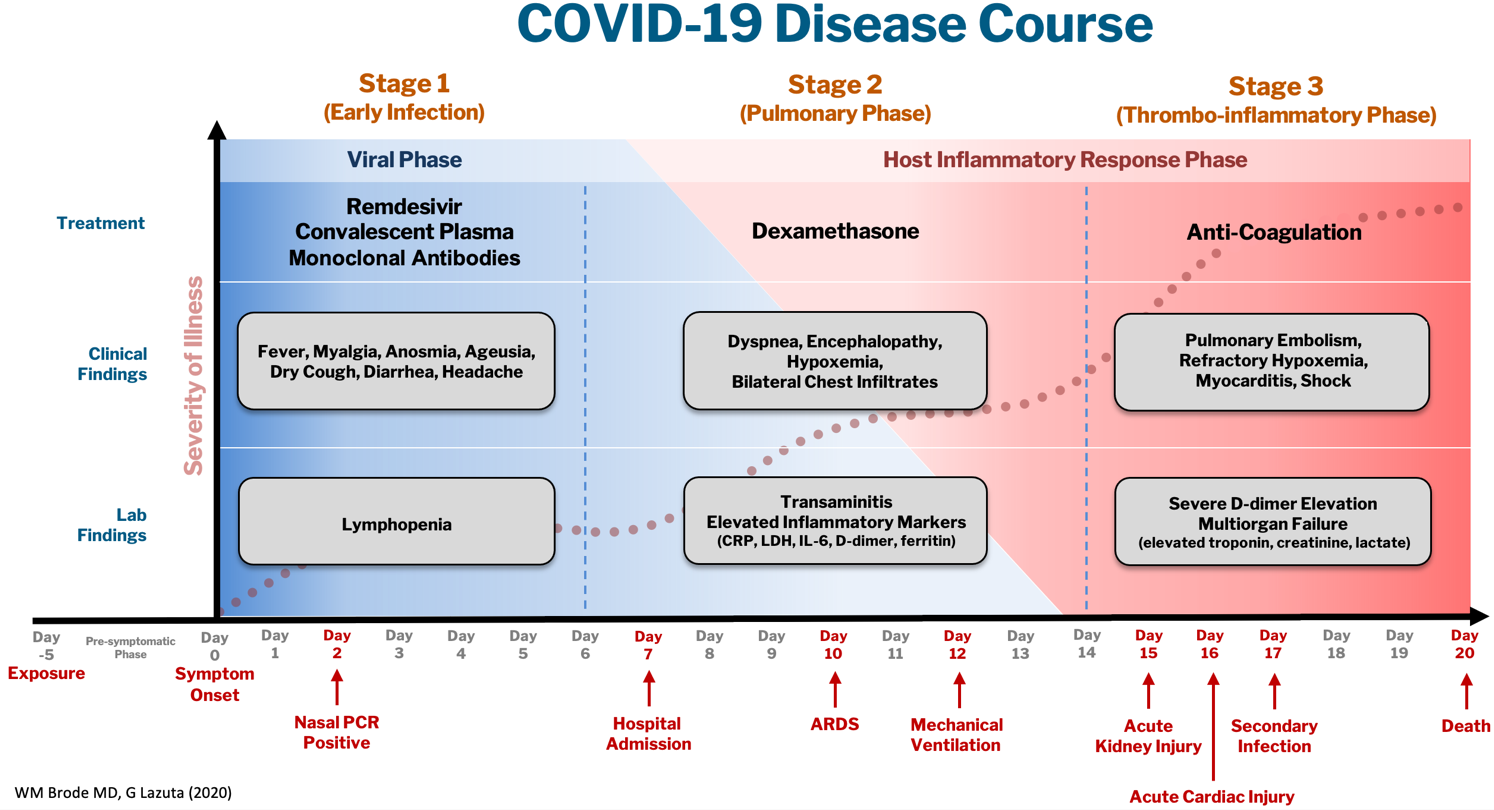
Stage 1 is the viral phase following a median 5-day pre-symptomatic phase from exposure; this is indistinguishable from an influenza-like illness with the typical fever, cough, GI symptoms, and the more specific anosmia, ageusia and orthostasis.
Stage 2 is the pulmonary phase where patients develop COVID-19 pneumonia and will have diffuse chest infiltrates on imaging. This stage usually represents the tail end of the viral phase prior to recovery, but for the ~15% of patients who present to the hospital needing admission because of hypoxemia (the definition of severe COVID-19, typically 5-7 days from symptom onset) this phase is characterized by elevated inflammatory markers and an exuberant host-immune response.
Stage 3 is the dreaded thrombo-inflammatory phase, which is a late manifestation usually >10 days from symptom onset and seems to be independent of viral replication. The morbidity and mortality associated with COVID-19 is likely a result of diffuse microthrombosis, and critical disease should no longer be thought of as a “cytokine storm,” but as life-threatening organ dysfunction caused by a dysregulated host response to infection. Unlike sepsis, the predominant pathology is not vasodilation and shock, but a hypercoagulable state with diffuse endothelial damage4,5.
Workup on presentation to the hospital should focus on identifying which phase of illness the patient is in, based on timing of symptom onset, inflammatory markers, and end-organ damage. CBC, CMP, D-dimer, troponin, and CRP are likely sufficient baseline labs in addition to a chest X-ray. There are many risk stratification tools, but to date, the 4C Mortality Score is recommended due to its large derivation cohort and reliance on only 8 practical variables6.
Remdesivir and convalescent plasma (CVP) disrupt viral replication in stages 1 and 2 of the illness. Remdesivir has shown efficacy reducing hospital length of stay and a small trend towards decreasing mortality, especially if given within 10 days of symptom onset, although its effectiveness in general use is very small, if it exists at all7,8. CVP efficacy has been disappointing and should not be the standard of care: multiple RCTs do not show any clinical benefit, although the Mayo Clinic registry data suggests that high-titer CVP given within 3 days from diagnosis decreases mortality compared to low-titer plasma9–11. Monoclonal antibodies are theoretically “supercharged” high-titer CVP, but are approved for outpatient use only. Trials for inpatients requiring oxygen were stopped due to futility.
Dexamethasone is the only treatment with a proven mortality benefit. The RECOVERY trial showed the greatest mortality benefit (NNT of 8) in mechanically ventilated patients > 7 days from symptom onset. While there is a benefit to patients requiring any oxygen (NNT of 30), early administration to patients in the viral phase is associated with higher mortality as corticosteroids can reduce viral clearance12. Corticosteroids should therefore be targeted to a therapeutic window to reduce the dysregulated host immune response and treat ARDS in phases 2 and 3.
Incidence of venous thromboembolism (VTE) increases linearly with disease severity (metanalysis showing a rate of 24% in the ICU13) and autopsy studies demonstrate diffuse microthrombosis even when VTE was not suspected5. Observational studies have shown VTE pharmacoprophylaxis reduces mortality, but the optimal agent, timing, and intensity of regimens is not clear14–16. Major society guidelines are equivocal on the use of anticoagulation, despite many institutions adopting higher prophylactic VTE doses for patients based on elevated D-dimer levels or disease severity. Anticoagulation trials in COVID-19 are ongoing, but for now intensified regimens vs. equipoise remain expert opinion only.
The most important treatment remains delivering oxygen with fidelity, correcting the much-observed “silent” or “happy hypoxemic”17. Given the high mortality associated with mechanical ventilation and that hypoxemia can be out of proportion to respiratory distress, arbitrary thresholds should not be used to decide when to intubate and instead should evaluate work of breathing, hypercapnia, mentation, or progression of end-organ damage rather than a single cutoff18. High-flow nasal cannula (HFNC) can correct severe hypoxemia in addition to self-proning, and while there is scant outcomes data for this strategy, it has been adopted widely as ICU capacity is strained nationally. A ventilator can add PEEP for alveolar recruitment or perform the work of breathing for a patient, but a patient will receive 100% FiO2 whether it is delivered through the nares on HFNC or 10 inches lower by an endotracheal tube.
In the absence of a single therapeutic cure or breakthrough, caring for a COVID-19 patient requires the hospital system to instead do a thousand things conscientiously and consistently. This is supportive care: most patients will get better with time and attentive evaluation for end-organ complications like myocarditis, encephalopathy, or pressure ulcers. It requires nursing to patient ratios that allows for this type of vigilance, with shared protocols, order sets, and close communication among team members that provides this support. The treatment of COVID-19 continues to evolve, but as we confront rising hospital volumes nationally, it is important to standardize care for patients throughout each of the 3 stages of illness until we find that single breakthrough.
References:
- Cummings, M. J. et al. Epidemiology, clinical course, and outcomes of critically ill adults with COVID-19 in New York City: a prospective cohort study. The Lancet (2020) doi:10.1016/S0140-6736(20)31189-2.
- Oudkerk, M. et al. Diagnosis, Prevention, and Treatment of Thromboembolic Complications in COVID-19: Report of the National Institute for Public Health of the Netherlands. Radiology 201629 (2020) doi:10.1148/radiol.2020201629.
- Siddiqi, H. K. & Mehra, M. R. COVID-19 illness in native and immunosuppressed states: A clinical–therapeutic staging proposal. J. Heart Lung Transplant. 39, 405–407 (2020).
- Connors, J. M. & Levy, J. H. COVID-19 and its implications for thrombosis and anticoagulation. Blood 135, 2033–2040 (2020).
- Ackermann, M. et al. Pulmonary Vascular Endothelialitis, Thrombosis, and Angiogenesis in Covid-19. N. Engl. J. Med. (2020) doi:10.1056/NEJMoa2015432.
- Knight, S. R. et al. Risk stratification of patients admitted to hospital with covid-19 using the ISARIC WHO Clinical Characterisation Protocol: development and validation of the 4C Mortality Score. BMJ m3339 (2020) doi:10.1136/bmj.m3339.
- Beigel, J. H. et al. Remdesivir for the Treatment of Covid-19 — Final Report. N. Engl. J. Med. 0, null (2020).
- Repurposed antiviral drugs for COVID-19; interim WHO SOLIDARITY trial results | medRxiv. https://www.medrxiv.org/content/10.1101/2020.10.15.20209817v1.
- Agarwal, A. et al. Convalescent plasma in the management of moderate covid-19 in adults in India: open label phase II multicentre randomised controlled trial (PLACID Trial). BMJ 371, (2020).
- Simonovich, V. A. et al. A Randomized Trial of Convalescent Plasma in Covid-19 Severe Pneumonia. N. Engl. J. Med. (2020) doi:10.1056/NEJMoa2031304.
- Joyner, M. J. et al. Effect of Convalescent Plasma on Mortality among Hospitalized Patients with COVID-19: Initial Three-Month Experience. medRxiv 2020.08.12.20169359 (2020) doi:10.1101/2020.08.12.20169359.
- The RECOVERY Collaborative Group. Dexamethasone in Hospitalized Patients with Covid-19 — Preliminary Report. N. Engl. J. Med. (2020) doi:10.1056/NEJMoa2021436.
- Porfidia, A. et al. Venous thromboembolism in patients with COVID-19: Systematic review and meta-analysis. Thromb. Res. 196, 67–74 (2020).
- Nadkarni, G. N. et al. Anticoagulation, Mortality, Bleeding and Pathology Among Patients Hospitalized with COVID-19: A Single Health System Study. J. Am. Coll. Cardiol. (2020) doi:10.1016/j.jacc.2020.08.041.
- Paranjpe, I. et al. Association of Treatment Dose Anticoagulation With In-Hospital Survival Among Hospitalized Patients With COVID-19. J. Am. Coll. Cardiol. 76, 122–124 (2020).
- Antithrombotic Therapy | Coronavirus Disease COVID-19. COVID-19 Treatment Guidelines https://www.covid19treatmentguidelines.nih.gov/adjunctive-therapy/antithrombotic-therapy/.
- Tobin, M. J., Laghi, F. & Jubran, A. Why COVID-19 Silent Hypoxemia is Baffling to Physicians. Am. J. Respir. Crit. Care Med. (2020) doi:10.1164/rccm.202006-2157CP.
- Berlin, D. A., Gulick, R. M. & Martinez, F. J. Severe Covid-19. N. Engl. J. Med. 0, null (2020).



Thanks
Carefully thought out presentation
Perhaps the best article written. It is sober in acknowledging the small effect size if any of most therapeutic treatment options besides steroid use in the inflammatory phase of COVID. Even in that case I have not seen such a trial replicated with concern for lack of clinical equipoise. Is CoViD ARDS different from other ARDS where steroid use has been controversial at best. Perhaps the one thing what I have learned is the desire to do something trumps the scientific method that is supposed to be used to confirm that “something” is truly efficacious.